The Phytochemical Landscape

Plants produce over 300,000 types of phytochemicals, and scientists are just beginning to realize that these unique compounds shape all life on Earth.
I started researching the phytochemical landscape last summer, but it's such an immense and extraordinarily complex topic that I've been reluctant to write about it. Even now, I can only offer the briefest sketch of some ways that the phytochemical landscape impacts all aspects of life as we know it.
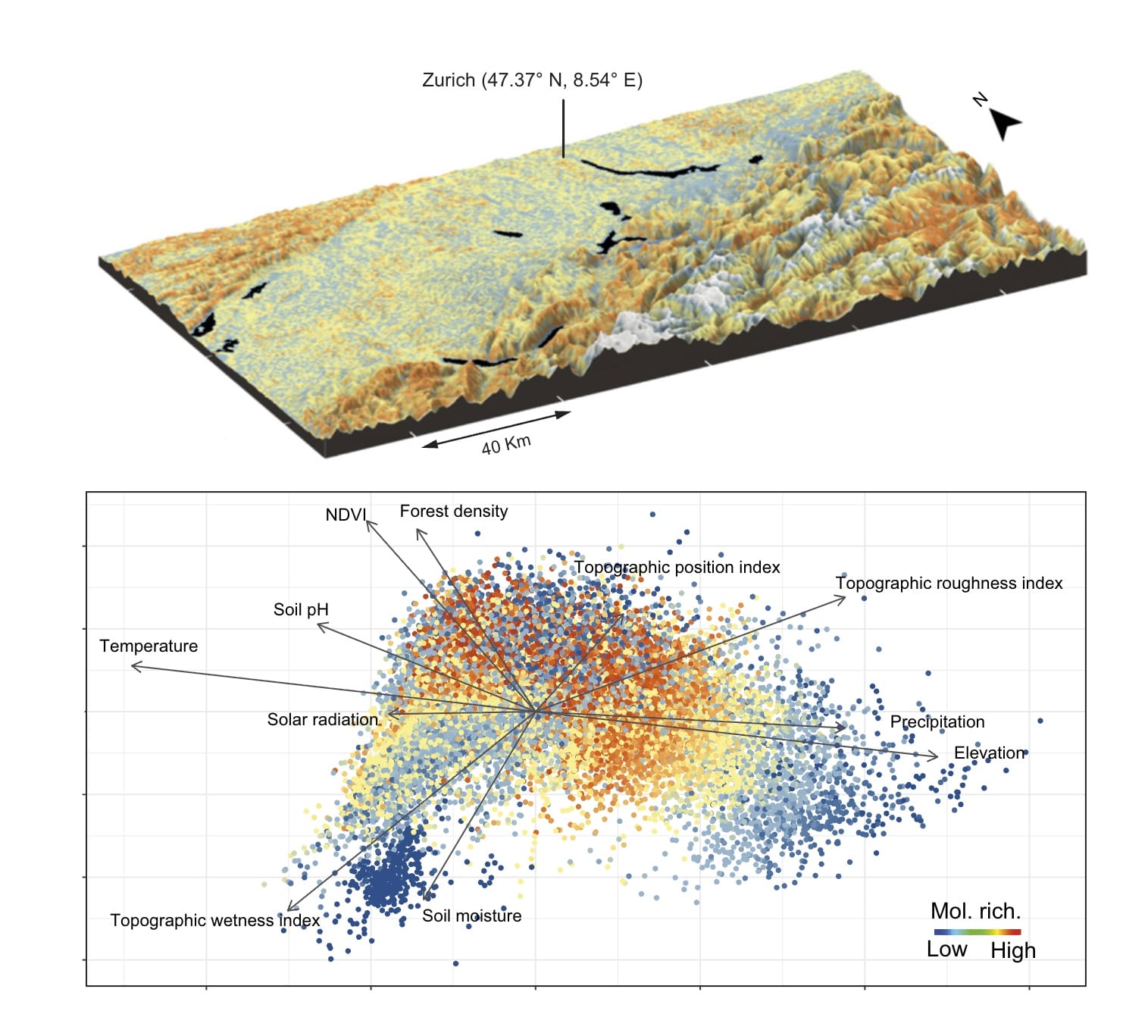
Basically, all life on Earth, and the distribution of all living things, is determined by the chemistry of plants. That makes sense, but the story is not quite so simple because phytochemicals shape life and life shapes phytochemicals in a multi-dimensional, ever-changing dance.
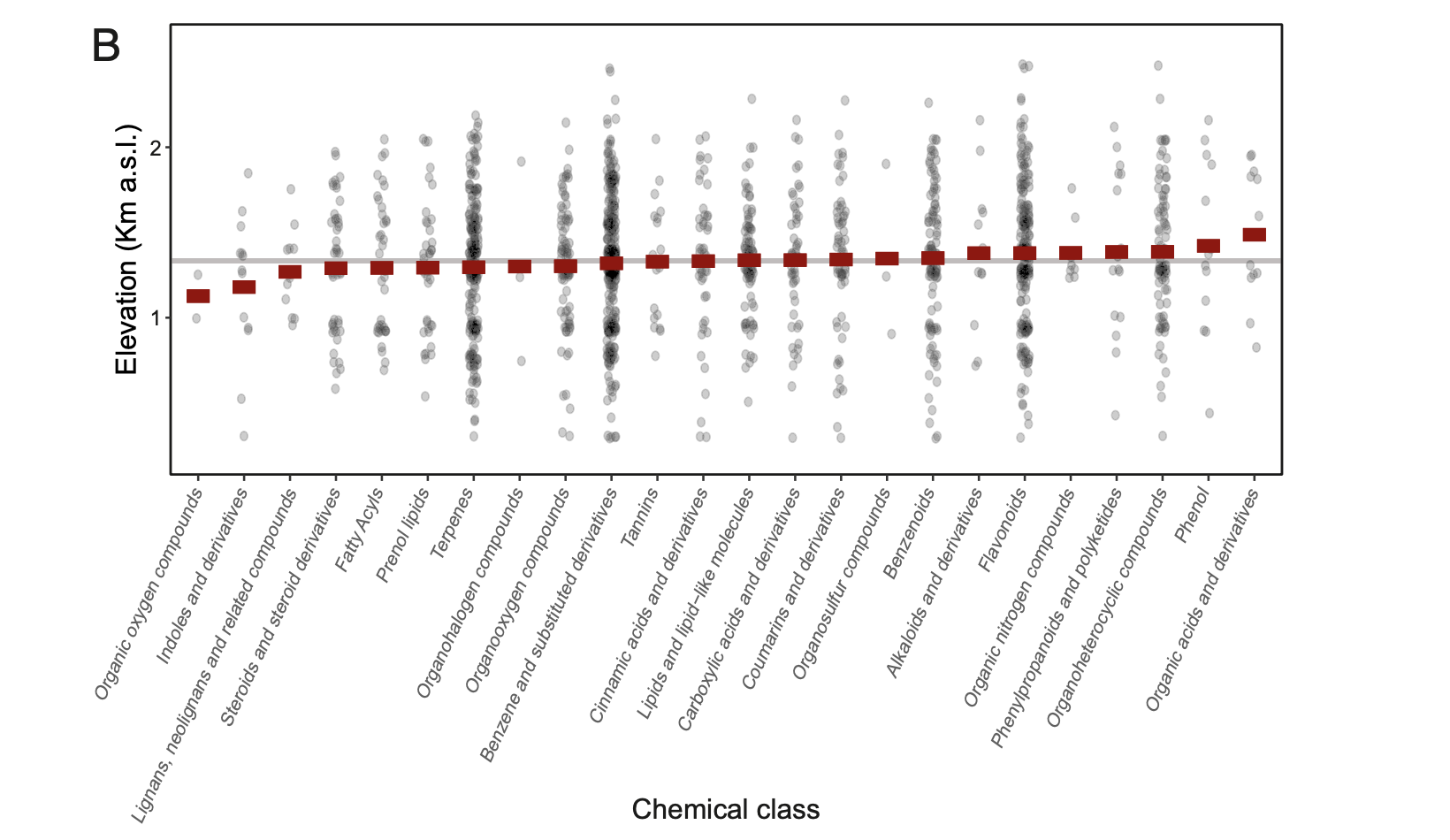
Ultimately, plants grow in different ways (with different chemical compositions) and in different places based on soil, sunlight, water, elevation, and nutrient availability, and then other organisms that are unique to each landscape, from fungi and bacteria to caterpillars and deer, eat these plants. Nutrients are then recycled back into the soil, which produces a new crop of plants that reflects the unique nutrients found in that soil.

What's amazing is that we can see these complex feedback loops play out across the landscape in ways that boggle the mind. For example, did you know that the chemistry of plants changes over the course of seconds, days, weeks, and years, and that the chemistry of plants varies significantly across the tissues of a single plant, across individual plants, across species, across landscapes, and across biomes? On top of that, a large ungulate like a deer takes approximately 10 million bites every year, and every bite is a separate decision about which plant, or which part of a plant, to eat based on the chemistry of the previous bite they've taken.

All of these very small shifts and decisions add up to create the large-scale patterns that we see every time we look at the natural world. For instance, plants growing on poor soils will protect themselves from herbivores with a very different chemistry than plants growing on nutrient-rich soils, and those plants are going to attract different types of herbivores, parasites, pathogens, and predators which will then nourish the soil in different ways and produce patches of different types of plants that we see when we look at the world.
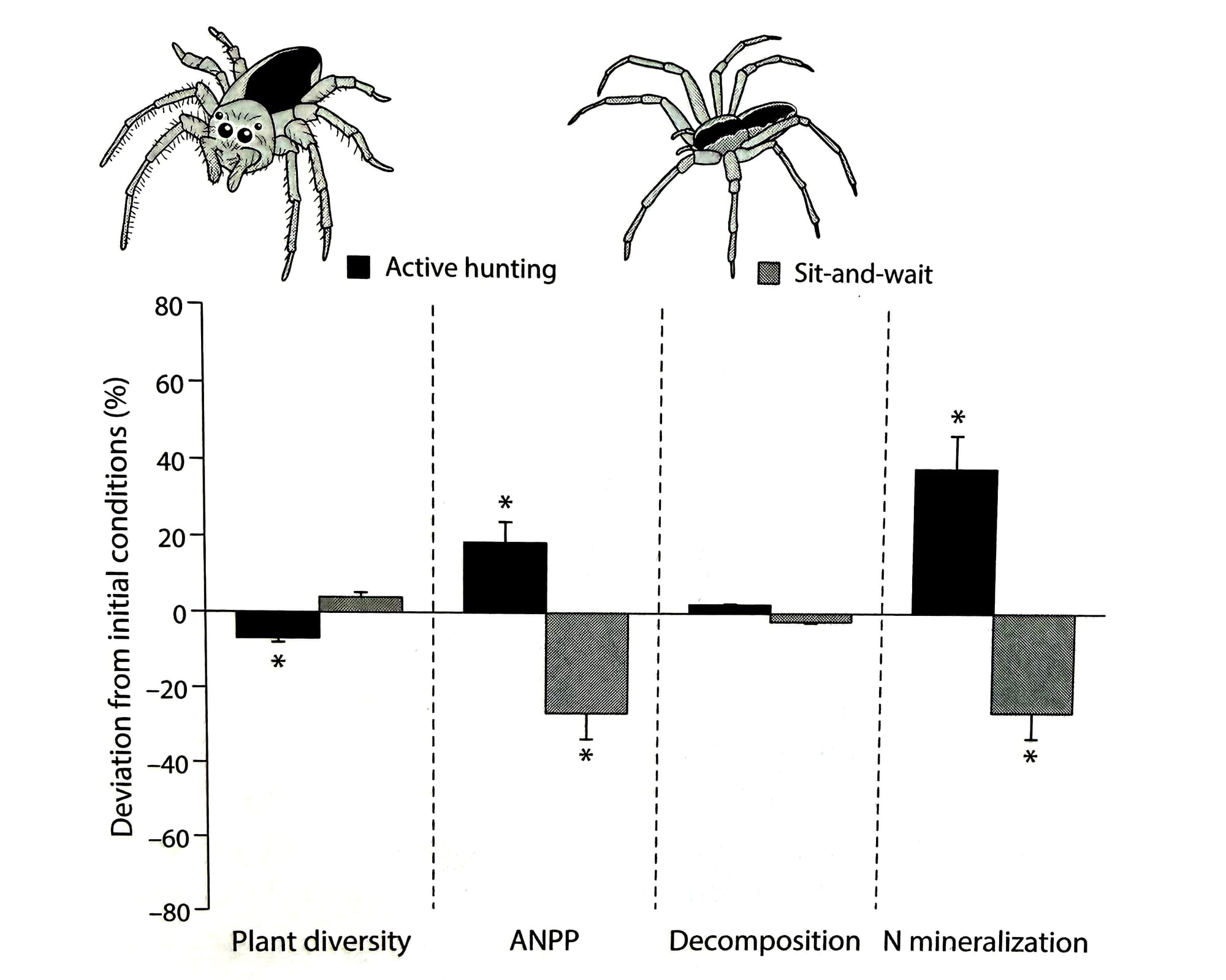
You can find examples of this happening all over the world; consider, for instance, giant termite mounds on the African savannah. Through their activity, termites concentrate nutrients in hot spots around their mounds, which in turn produces nutrient-rich plant communities that attract large grazing mammals, who then enrich the soil with their feces and fuel the growth of even more types of plants.

Or consider that killing wolves in Alaska leads to increased numbers of moose, who then eat greater numbers of deciduous plants that are replaced by flammeable conifers, leading to bigger and hotter fires that alter soil chemistry and lead to further changes in plant communities.
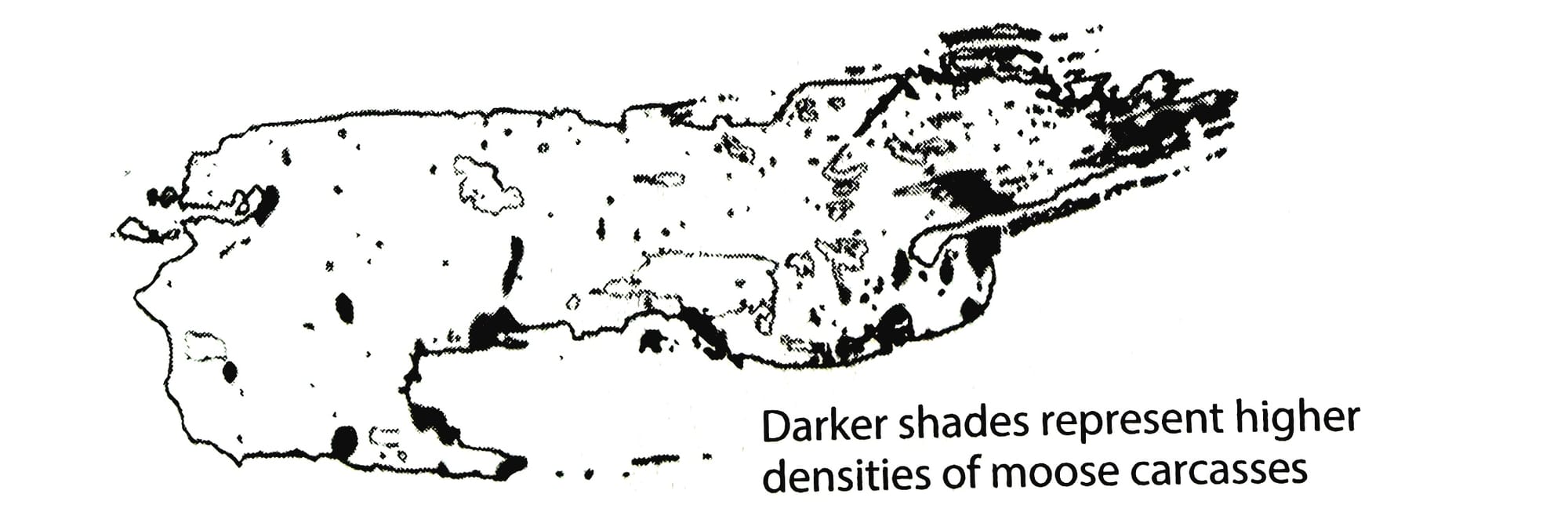
In other words, plant chemistry directly impacts the distribution, behavior, and abundance of plant-eating animals, which shapes the distribution, behavior, and abundance of predators. These factors then determine which nutrients are returned to the soil, how fast decomposition proceeds, how nitrogen is recycled, how carbon is utilized, and countless other aspects of soil ecology. Furthermore, all these dynamics are bi-directional, working up and down the chain of causation in endless, ever-changing loops of energy.
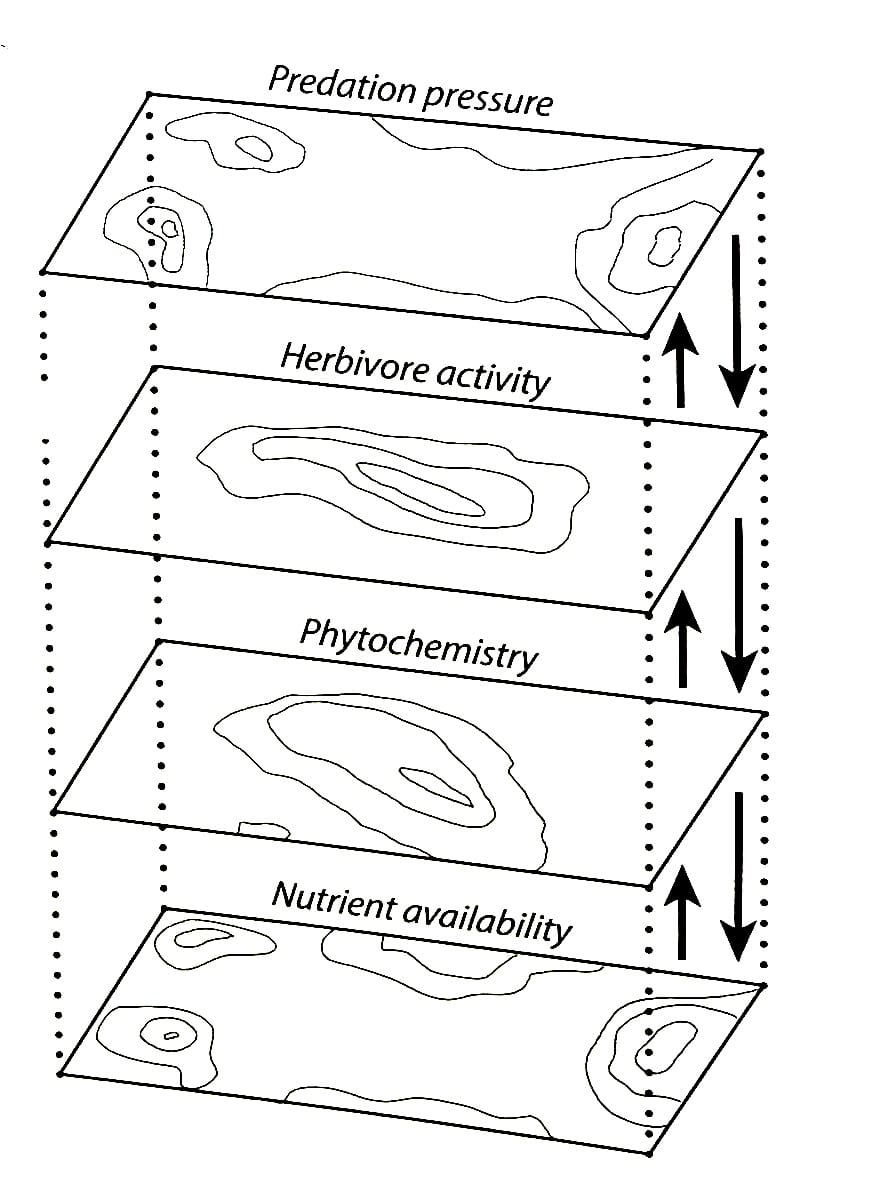
One key understanding that scientists are taking from the raft of new studies in this field is that species exist in complex webs of synergistic relationships. When a species is removed, or goes extinct, long-term changes in plant communities and soil ecology expand outward in profound ways that no one realized before.
Additional Resources:
Today's topic was inspired by a fascinating book called The Phytochemical Landscape by Mark Hunter. Unfortunately, this is an exceedingly dense book with a bibliography that spans 69 pages, but if you're willing to tackle it, the book is filled with hundreds of case studies and astonishing insights that were new to me.
For the extra-nerdy, I also found a link to the full text of a book called Plant Secondary Metabolism. A book like this is better used as a reference handbook, but the secondary compounds in plants is an incredible thing to study because they explain so much about the world around us.
And, one thing that makes secondary compounds so interesting is their medicinal properties. Not only does this matter to humans, but flies, moths, ants, and bees have all been documented using phytochemicals as medicines. If you know someone who is an herbalist, they might enjoy this paper Modes of Action of Herbal Medicines and Plant Secondary Metabolites.

Member discussion